2025-04-19: addendum-main forces in proteins
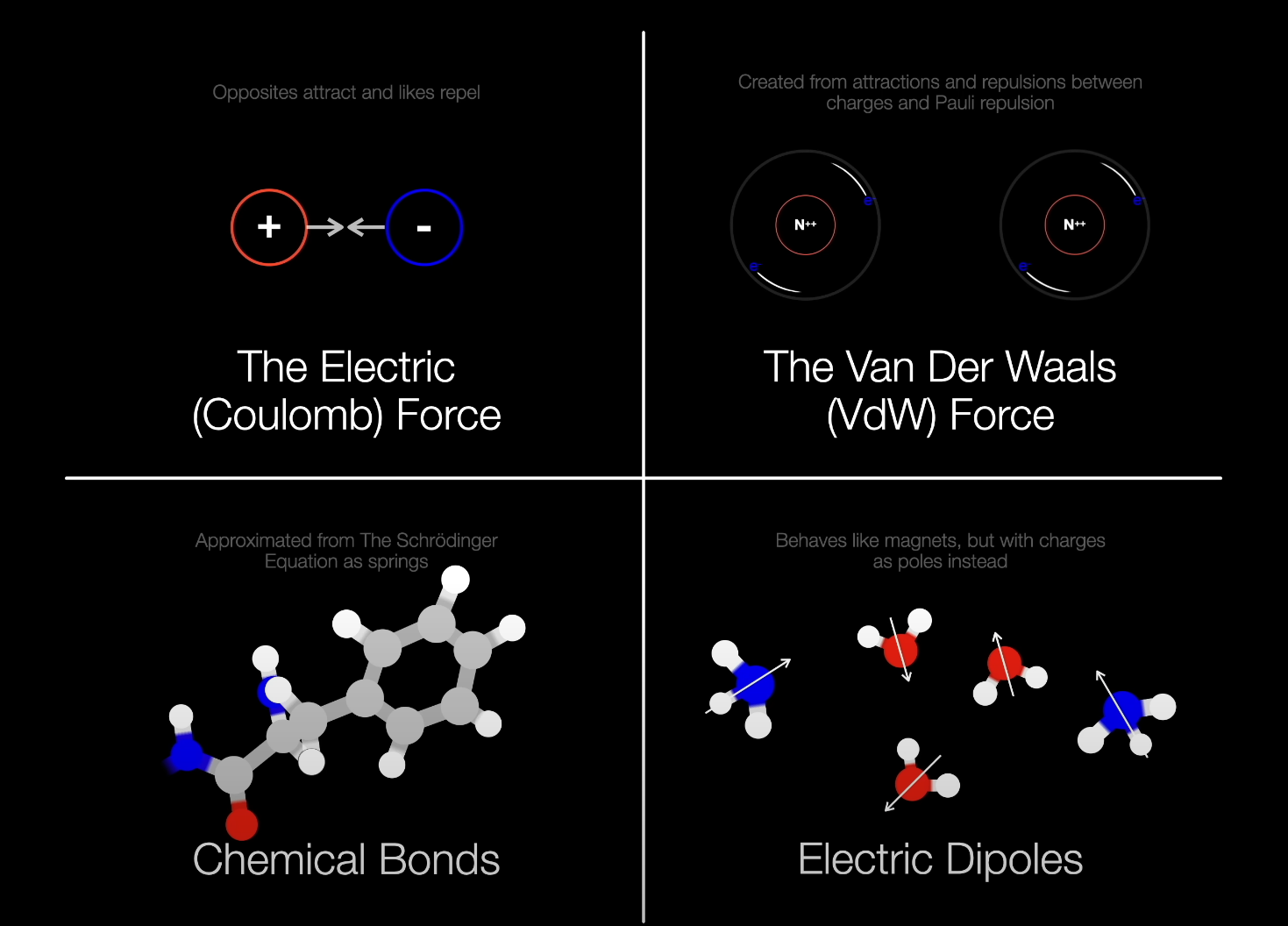
2024-09-10: First draft
source: https://youtu.be/EweuU2fEgjw?si=mEWLvHNat4fdqtEy
Introduction to Proteins
- Proteins are polymers of amino acids, also known as polypeptides.
- They are the most diverse type of biomolecule in the body , performing various functions:
- Enzymes: Catalyze chemical reactions.
- Receptors: Control signaling in the body.
- Hemoglobin: Carries oxygen in the bloodstream.
- Muscle and Organ Tissue: Provides structure and mobility.
Formation of Peptide Bonds
-
Peptide Bonds: Formed through dehydration reactions between amino acids.
- Example: Two glycine units form a peptide bond, losing a water molecule in the process.
-
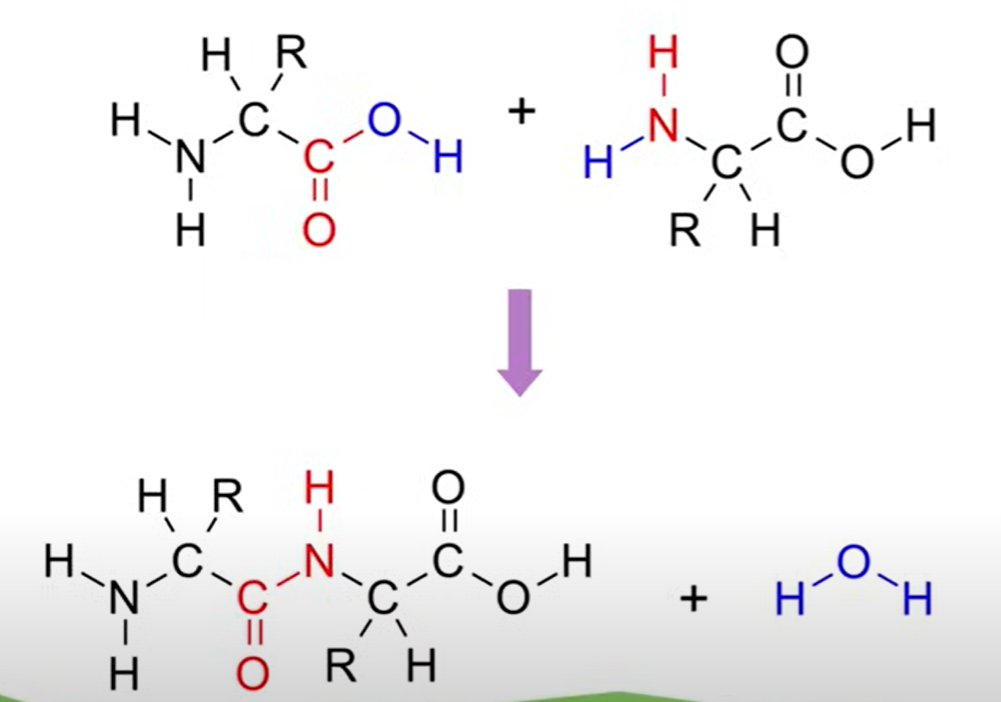
- This results in an amide functional group, connecting amino acids.
- Example: Two glycine units form a peptide bond, losing a water molecule in the process.
-
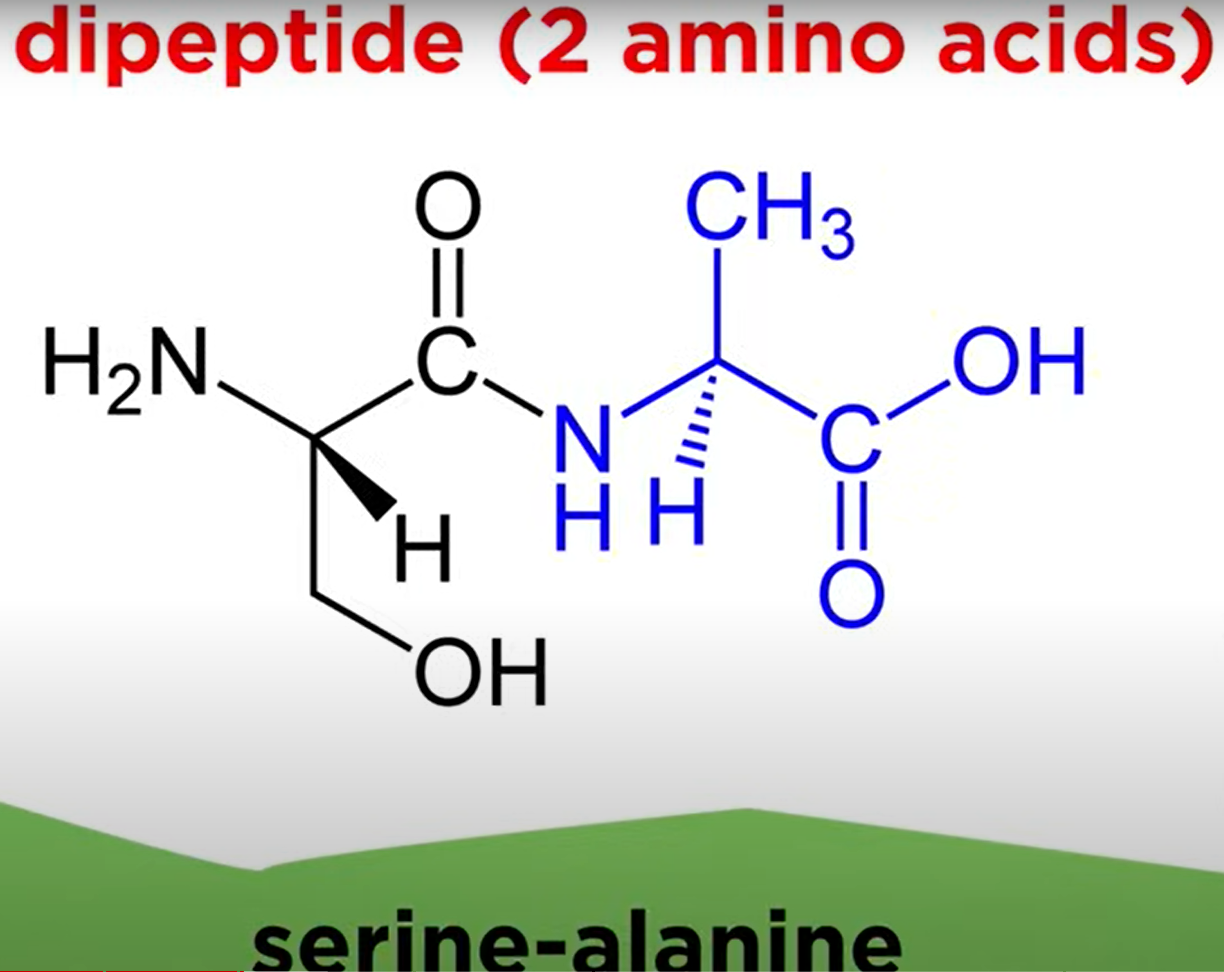
Types of Peptides
- Dipeptide: Two amino acids linked together.
- Oligopeptide: 3-10 amino acids.
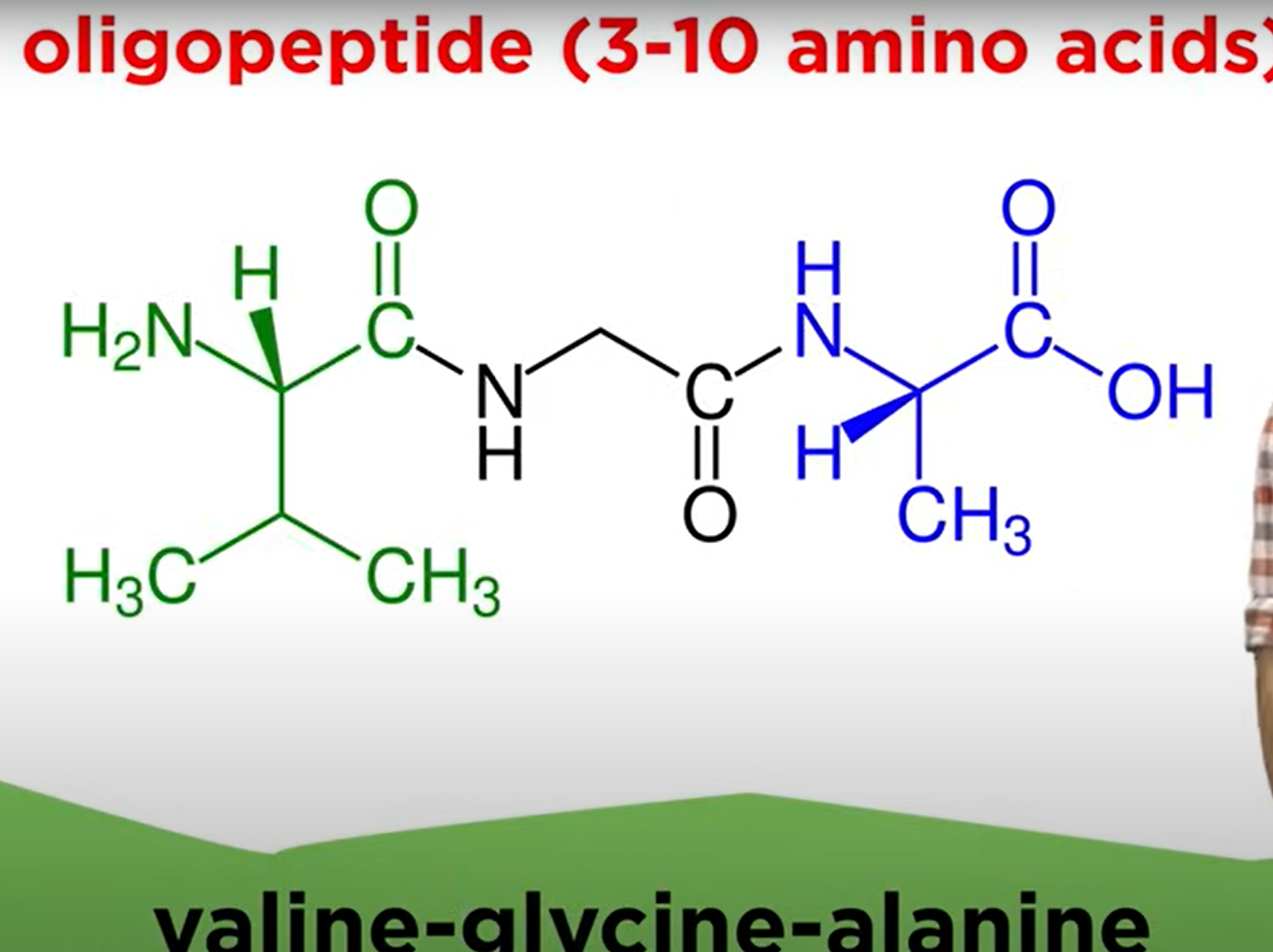
- Polypeptide: More than 10 amino acids, often around 300-1000 amino acids in functional proteins.
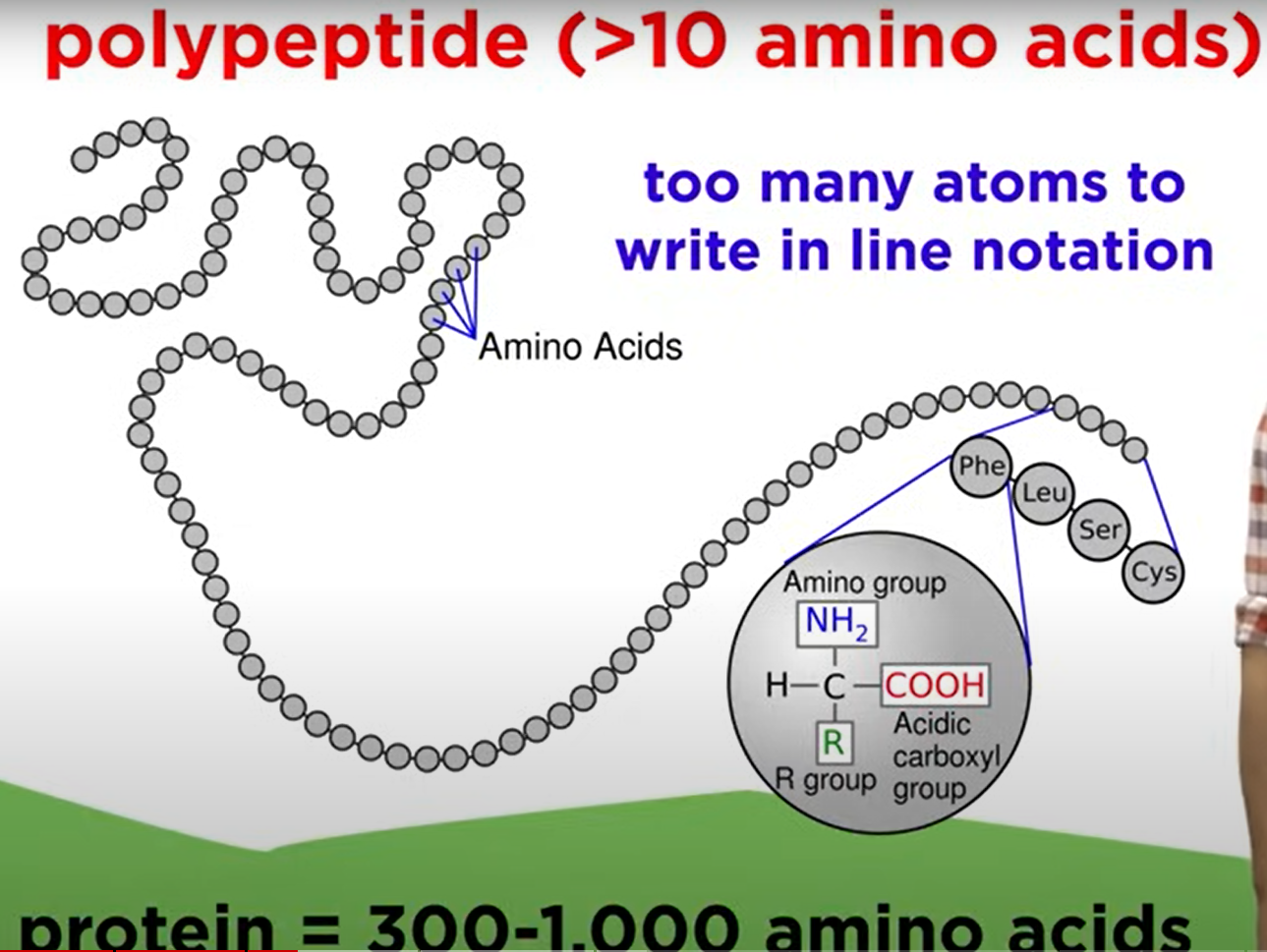
Protein Terminology
N-Terminus:
- End of the chain with the amino group.
C-Terminus:
End of the chain with the carboxyl group.
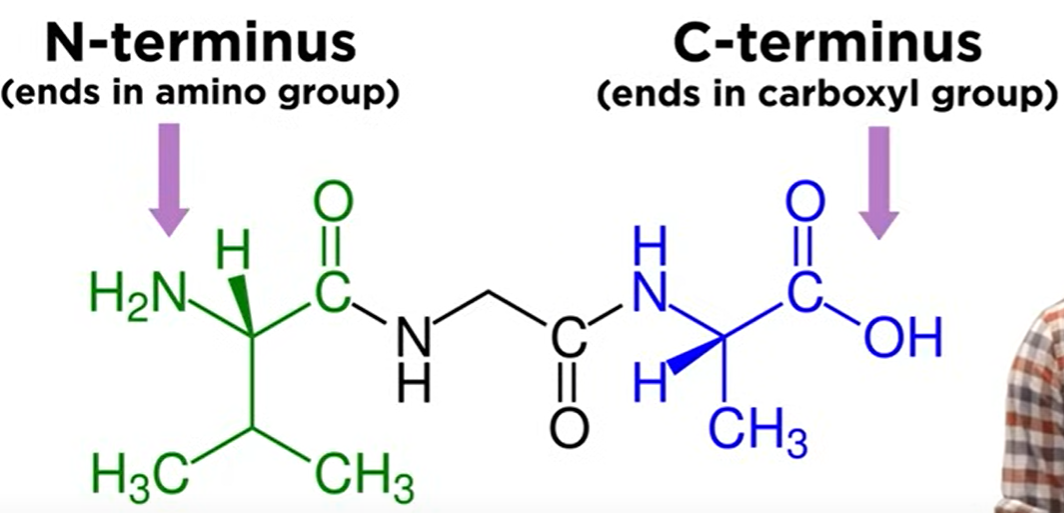 - By convention, N-terminus is written on the left and C-terminus is written on the right in molecular diagrams.
- By convention, N-terminus is written on the left and C-terminus is written on the right in molecular diagrams.
Residue:
Each monomeric unit in the polypeptide.
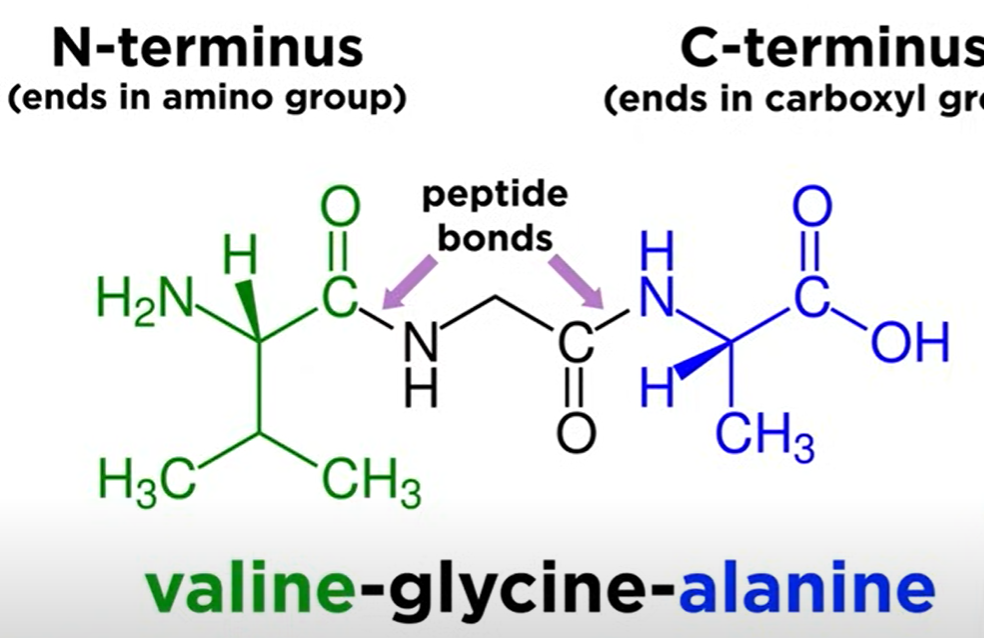 Each monomeric unit in the polypeptide is called residue, which can be differentiated by polypeptide bonds. In the example above there are three residues: valine, glycine and alanine.
Each monomeric unit in the polypeptide is called residue, which can be differentiated by polypeptide bonds. In the example above there are three residues: valine, glycine and alanine.
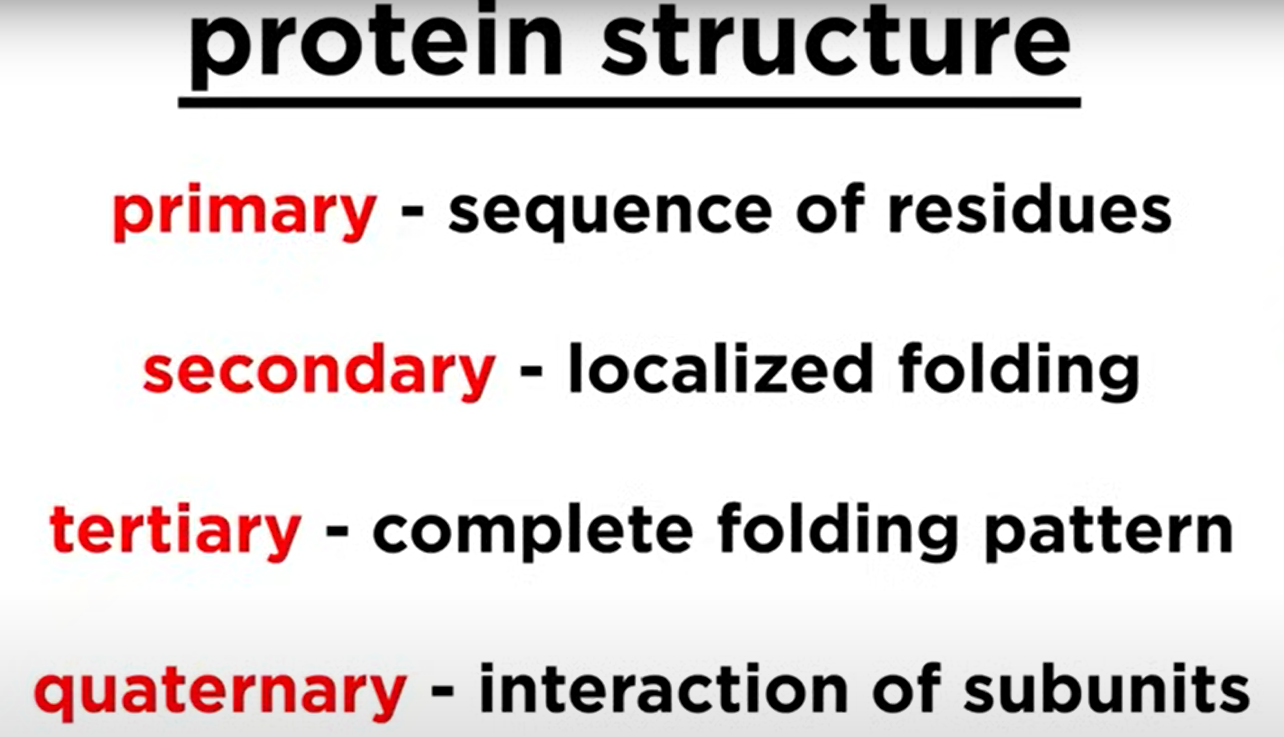
Protein Structure Hierarchy
Primary Structure
- Primary Structure: Sequence of amino acids.
- Represented using three-letter or one-letter codes.
 - Sets of abbreviations of amino acids in both three-letter-notation and one-letter-notation
- Sets of abbreviations of amino acids in both three-letter-notation and one-letter-notation

- Represented using three-letter or one-letter codes.
- The sequence determines how the protein will fold.
Secondary Structure
- Secondary Structure: Localized conformations of the polypeptide backbone to minimize the net electrostatic potential and hydrogen bond potential.
#beta-pleated-sheet alpha-helix
- Beta-Pleated Sheet: Backbone extends and turns back, forming dipole-dipole interactions.
- Alpha Helix: Backbone forms a spiral with 3-4 amino acids per turn, R groups pointing outward.
-
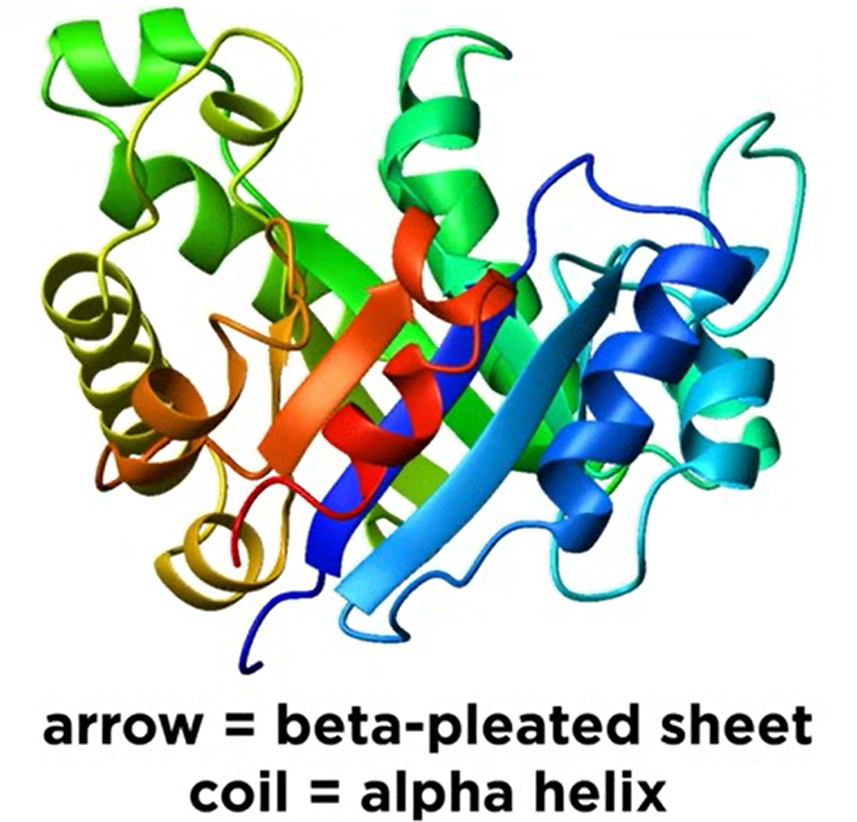
Tertiary Structure
- Tertiary Structure: Further folding of the polypeptide chain into its 3D structure.
- Hydrophobic Side Chains: Tend to be inside the protein.
- Hydrophilic Side Chains: Tend to be on the protein surface. They usually have net electrostatic charge and dipole moment to maximize the dipole-dipole Interactions and ion-dipole Interactions with the surroundings.
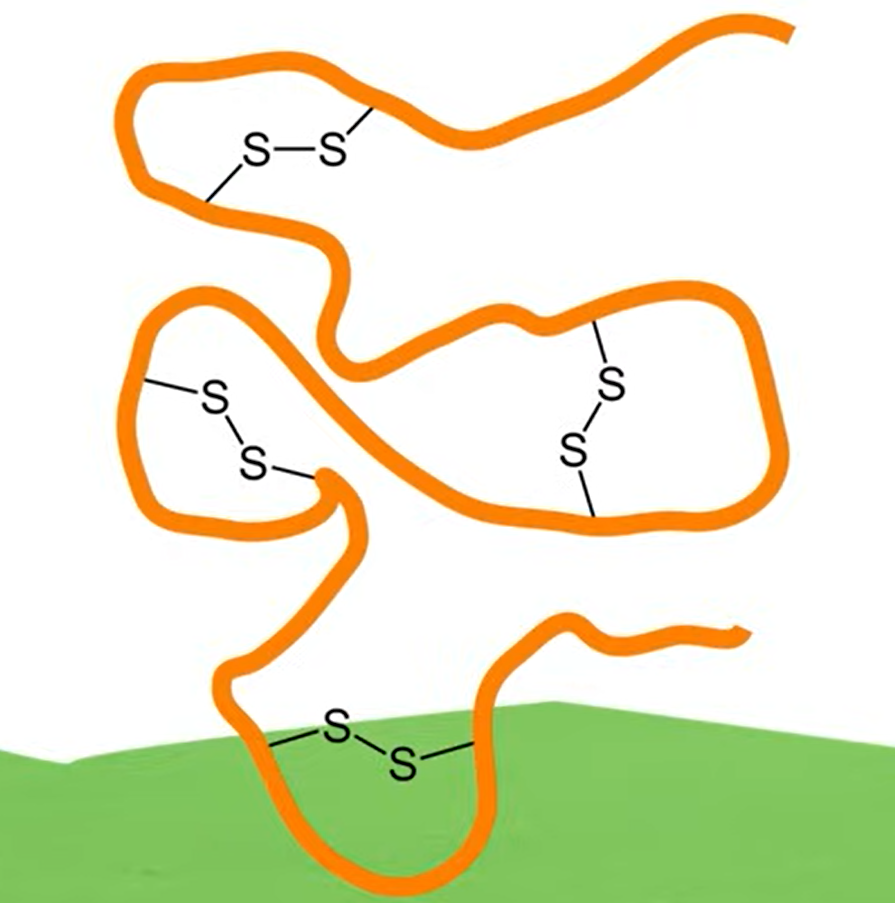
- Disulfide Bonds: Covalent linkages between cysteine residues.
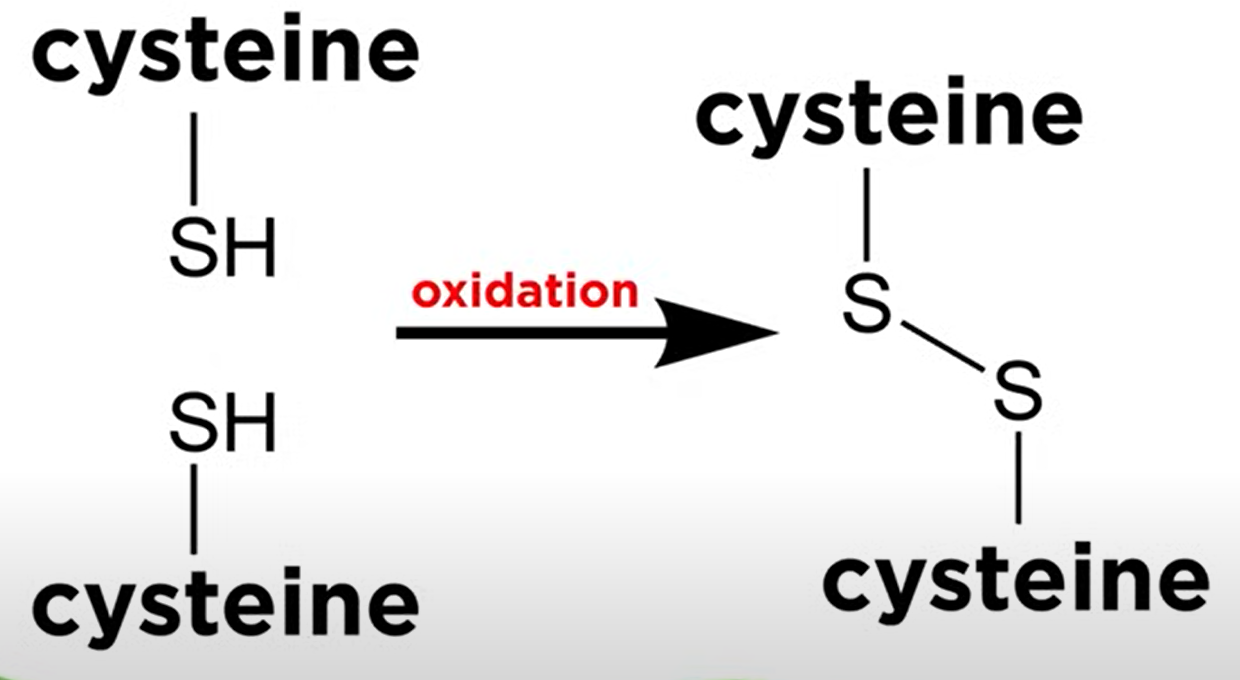 These covalent linkage also help maintain the folded structure of the protein:
These covalent linkage also help maintain the folded structure of the protein:
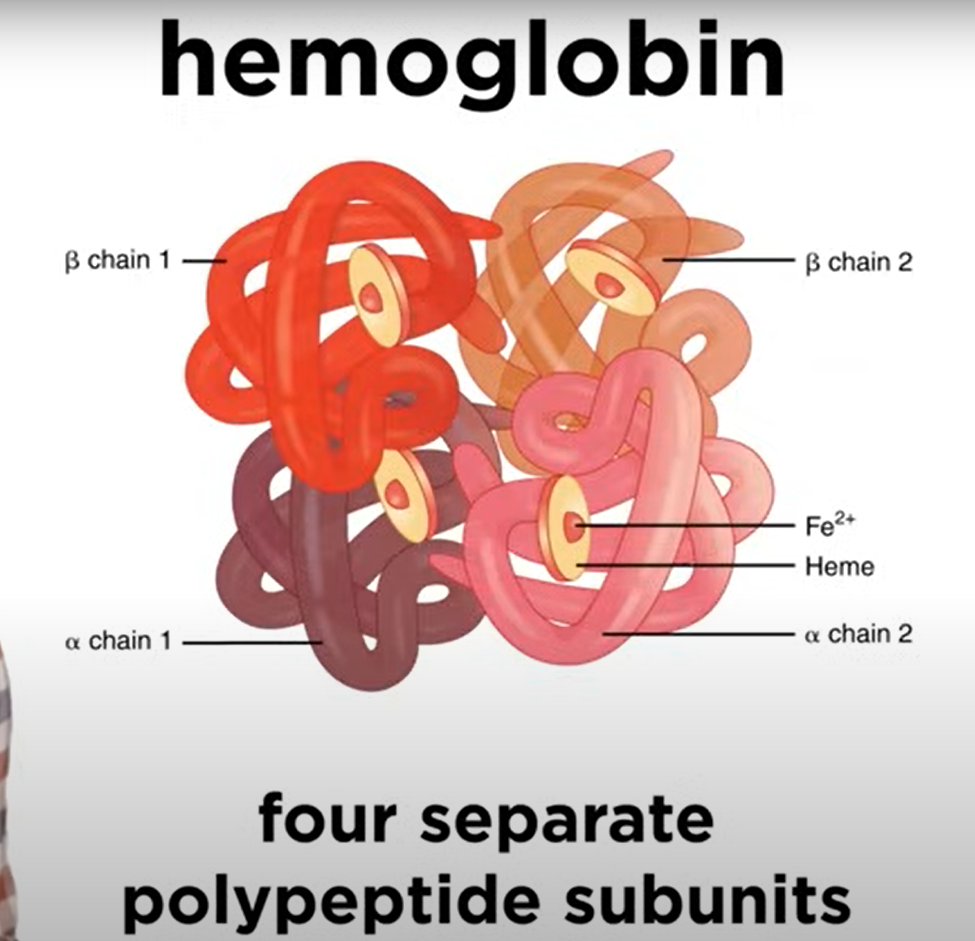
Quaternary Structure
- Quaternary Structure: Arrangement of multiple polypeptide subunits.
- Subunits are not covalently bound but interact electrostatically.
- Example: Hemoglobin consists of four polypeptides.
Importance of Protein Structure
- Sickle Cell Disease: A mutation in the primary structure (glutamic acid to valine) changes the folding pattern, leading to misshapen red blood cells.
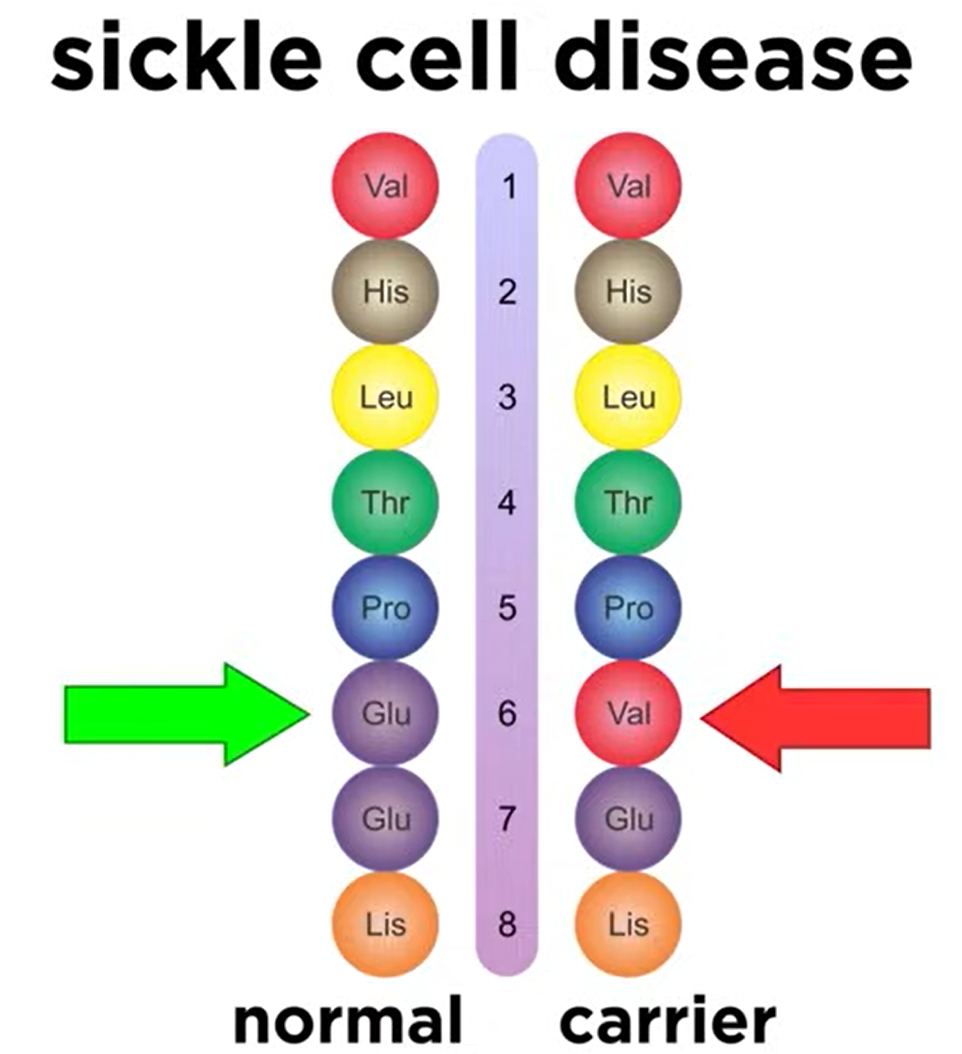
- Understanding protein structure is crucial for studying health and disease.
Conclusion
- Understanding the hierarchy of protein structure (primary, secondary, tertiary, quaternary) is essential for grasping their function and role in biological processes.
For further learning, follow Professor Dave’s tutorials on his YouTube channel.